About InfCareHIV
The InfCareHIV cohort was established in the counties of Stockholm and Gothenburg, Sweden, in 2003. Since 2008 the cohort has had complete national coverage, including all Sweden’s clinical HIV centres.
More than 99% of all people diagnosed with HIV in Sweden are included in InfCareHIV, a number that has been validated repeatedly against among others the Public Health Authority.
Variables collected at enrolment include sex at birth, gender identity, country of birth, suspected mode of HIV transmission, date of any last HIV-negative test, date of first positive HIV-test, and suspected country for HIV transmission. Data collected/updated at each follow-up visit include all HIV-RNA-levels, HIV drug resistance (including the viral sequences) results and CD4+ cell counts. Start and and stop dates of antiretroviral therapy and the reason for any change of drug regimens. Serological and virological data on co-infection with hepatitis C and B virus, weight, date and type of eventual AIDS events, date and cause of death are also included.
Validation of data is performed regularly on national level by the use of a Data Quality Index, with the possibility to validate also on unit-level only. Data Quality Index for InfCareHIV is currently at 4.9 on a 0-5 scale.
At time of HIV diagnosis the person diagnosed is asked consent for inclusion in InfCareHIV and may decline without giving any reasons. Participants always have the right to exit InfCareHIV. Participants can request an extract on their data from the register, free of charge, in accordance with the European General Data Protection Regulation (GDPR 2016/679) and the Swedish Data Protection Act (2018:218).
Decision support system
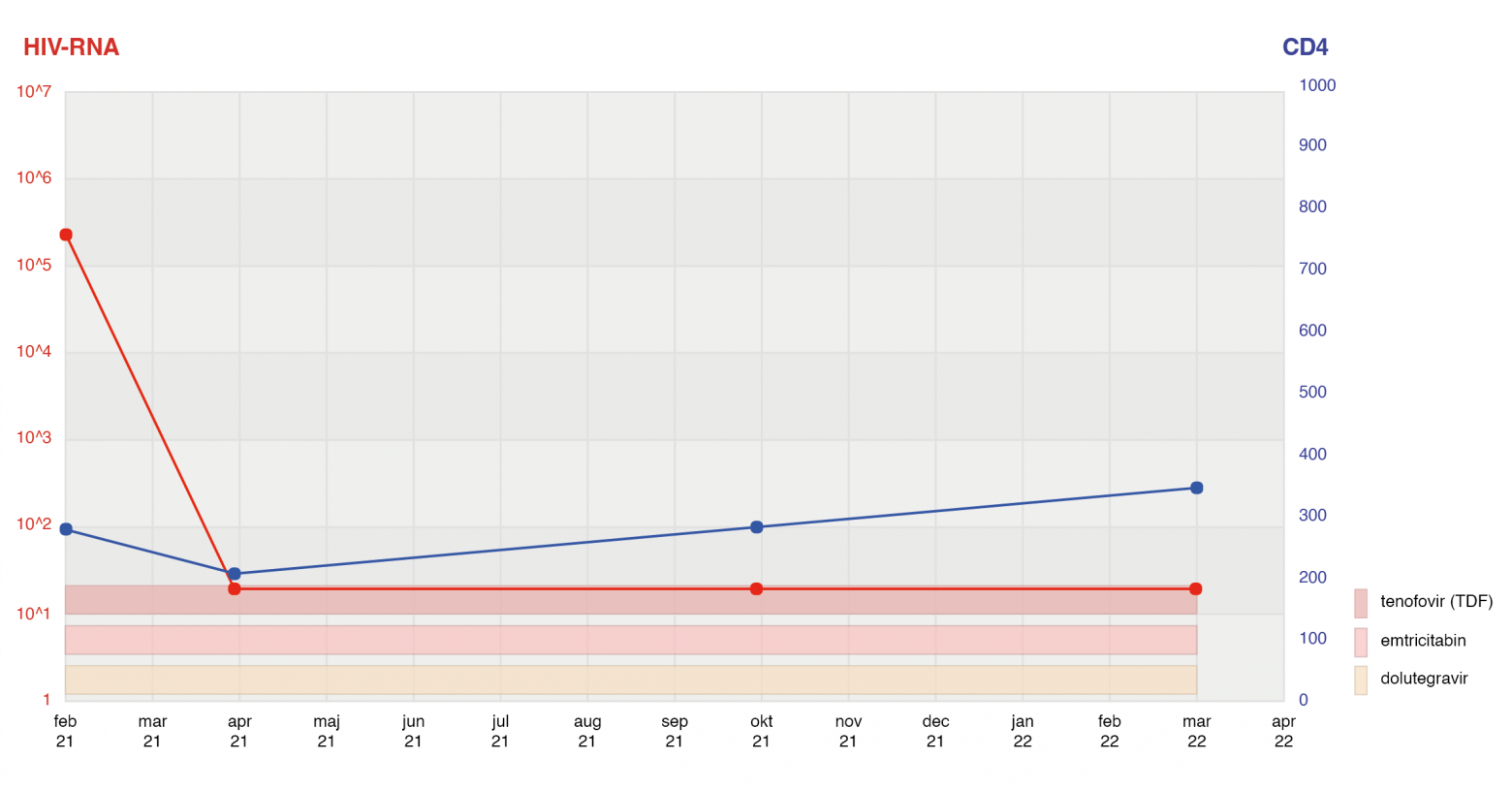
The mainstay of InfCareHIV is the clinical decision support system. Data on HIV-RNA level, CD4 count, drug resistance, and drug treatment are presented through a graphical system.
Quality Register
InfCareHIV also functions as a National Quality Register. The aim of a quality register is to systematically and continuously develop and safeguard quality of care.
Health-related quality of life
The aim is for all PWH to be invited, once annually, to answer a self-reported health questionnaire that assesses patient-reported outcome measures (PROMs), and patient-experienced measures (PREMSs). The questionnaire forms the basis for the patient consultation with nurse or doctor.
Research
InfCareHIV provides a unique base to study long-term treatment, comorbidity and health-related quality of life in people with HIV in Sweden. Ethical permission from the Swedish Ethical Review Authority is needed before applying to the InfCareHIV steering committee for access to anonymized data for research.